Header home
Le Conseil de l’Europe a 70 ans
1949 - 2019The Council of Europe at 70
1949 - 2019Asset Publisher
Asset Publisher
.jpg)
Statute of the Council of Europe is signed in London
United KingdomThe Treaty of London is signed by Belgium, Denmark, France, Ireland, Italy, Luxembourg, Netherlands, Norway, Sweden and the United Kingdom, founding the Council of Europe.
Read more
Committee of Ministers' first meeting
FrancePaul-Henri Spaak chairs first session of the Council of Europe Committee of Ministers.
Read more
Greece joins
GreeceGreece becomes the 11th member State of the Council of Europe.
Read more
Council of Europe gets its first Secretary General
FranceJacques Camille Paris becomes the first Secretary General.
Read more
Iceland joins
IcelandIceland becomes the 12th member State of the Council of Europe.
Read more
Turkey joins
TurkeyTurkey becomes the 13th member State of the Council of Europe.
Read more
Federal Republic of Germany joins the Council of Europe
GermanyThe Federal Republic of Germany joins the Council of Europe, becoming the 14th member State.
Read more
Winston Churchill speaks at Parliamentary Assembly.
United KingdomWinston Churchill, Prime Minister of the United Kingdom from 1940 to 1945 and again from 1951 to 1955, addresses the Parliamentary Assembly.
Read more
Human Rights Convention signed in Rome
ItalyThe European Convention on Human Rights is signed in Rome marking a new era of respect for human rights in Europe.
Read more
Cooperation begins with the United Nations
A fairly limited working agreement is signed between the General Secretariat of the Council of Europe and the Secretariat of the United Nations, allowing relations between the two organisations to develop.
Read more
The Organisation's status is recognised
All CountriesGeneral agreement on privileges and immunities of the Council of Europe enters into force.
Read more
Human Rights Convention goes live
All CountriesEntry into force of the European Convention on Human Rights.
Read more
Second Secretary General takes office
FranceLéon Marchal becomes the second Secretary General.
Read more
First art exhibition in Brussels
BelgiumThe first Council of Europe art exhibition "Humanist Europe" / "L'Europe Humaniste" opens at the Palais des Beaux-Arts in Brussels.
Read more
Celebrating cultural diversity
FranceThe European Cultural Convention is opened for signature, aiming to foster understanding and promote cultural diversity.
Read more
A flag for Europe
FranceAdoption of the European Flag by the Council of Europe's Committee of Ministers.
Read more
Austria joins
AustriaAustria becomes the 15th Council of Europe member State.
Read more
Development Bank begins its work
FranceThe Council of Europe Resettlement Fund is set up (today the Council of Europe Development Bank) to provide support for social projects.
Read more
Hungarian Uprising
HungaryStudent protests begin the Hungarian uprising to challenge Soviet control.
Read more
Steps towards local democracy
FranceFirst session of the Conference of Local Authorities of Europe (today the Congress)
Read more
Third Secretary General takes office
ItalyLodovico Benvenuti becomes the third Secretary General.
Read more
Israeli Knesset granted observer status
IsraelThe parliament of Israel, the Knesset, is granted observer status with the Parliamentary Assembly.
Read more
Easier personal travel between countries
All CountriesEuropean Agreement on Regulations governing the Movement of Persons between Member States of the Council of Europe enters into force.
Read more
Human rights court is set up
FranceFirst members of the European Human Rights Court elected by the Council of Europe Consultative Assembly.
Read more
Court gets first President
United KingdomLord (Arnold Duncan) McNair becomes the first President of the European Human Rights Court.
Read more
First Court session
FranceEuropean Human Rights Court holds first session.
Read more
Refugees allowed to travel
All CountriesRefugees can move from one country to another thanks to an agreement on abolition of visas.
Read more
First human rights judgment
FranceThe European Human Rights Court delivers its first judgment following an individual application: Lawless v. Ireland.
Read more
Cyprus joins
CyprusCyprus becomes the 16th member State to join the Council of Europe.
Read more
A safeguard for social rights
ItalyEuropean Social Charter is adopted, safeguarding the economic & social rights of citizens.
Read more
Looking after the property of hotel guests
FranceOpening of the Convention on the Liability of Hotel-keepers concerning the Property of their Guests
Read more
Switzerland joins
SwitzerlandSwitzerland joins the Council of Europe becoming the 17th member State.
Read more
Fourth Secretary General takes office
United KingdomPeter Smithers becomes the fourth Secretary General.
Read more
A day for Europe
All CountriesThe Committee of Ministers decides that 5 May, the anniversary of the founding of the Council of Europe, should be celebrated as Europe Day.
Read more
Medicines made safe
FranceThe European Pharmacopoeia is launched to ensure safe medicine for all.
Read more
Malta joins
MaltaMalta becomes the 18th member State to join the Council of Europe.
Read more
Court gets second President
FranceRené Cassin becomes the second President of the European Human Rights Court.
Read more
Europe and art meet in Stockholm
Sweden"Christina, Queen of Sweden: a personality of European civilisation", the eleventh Council of Europe Art Exhibition opens at the Stockholm Nationalmuseum.
Read more
Protection of adopted children
FranceOpening of the European Convention on the Adoption of Children
Read more
Prague Spring
Czech RepublicThe Soviet Union and other Warsaw Pact members send armed forces into Czechoslovakia to suppress the mass protests and put an end to liberal reforms.
Read more
Court gets third President
BelgiumHenri Rolin becomes the third President of the European Human Rights Court.
Read more
Fifth Secretary General takes office
AustriaLujo Tončić-Sorinj becomes the fifth Secretary General.
Read more
Greece withdraws
GreeceGreece withdraws from the Council of Europe to avoid expulsion after a military coup.
Read more
Holy See becomes observer
Holy SeeThe Holy See is granted observer status with the Council of Europe.
Read more
Court gets fourth President
United KingdomSir Humphrey Waldock becomes the fourth President of the European Human Rights Court.
Read more
An anthem for Europe
FranceThe Council of Europe chooses Beethoven's "Ode to Joy", the fourth movement of the composer's ninth symphony, as the European Anthem.
Read more

A uniform mortuary "laissez-passer"
FranceAgreement on the Transfer of Corpses simplifies formalities required for the international transfer of corpses, through a uniform mortuary "laissez-passer".
Read more
Portugal's peaceful coup
PortugalBeginning as a military coup, Portugal's Carnation Revolution brings an end to dictatorship.
Read more
Court gets fifth President
ItalyGiorgio Balladore Pallieri becomes the fifth President of the European Human Rights Court.
Read more
Sixth Secretary General takes office
GermanyGeorg Kahn-Ackerman becomes the sixth Secretary General to take office.
Read more
Greece re-joins
GreeceGreece returns to the Council of Europe after the end of military dictatorship and the restoration of democracy.
Read more
Franco dies
SpainThe death of dictator General Franco opens the way for Spain's transition to democracy.
Read more
Portugal joins
PortugalPortugal is the 19th member State to join the Council of Europe.
Read more
Palais de l'Europe opens
FranceInauguration by French President, Giscard d'Estaing of the Palais de l'Europe, the home of the Council of Europe.
Read more
Spain joins
SpainSpain becomes the 20th member State to join the Council of Europe.
Read more
Court's first inter-state judgment
FranceFirst judgment given by the European Human Rights Court concerning an inter-state case - Ireland v. United Kingdom.
Read more
Liechtenstein joins
LiechtensteinLiechtenstein becomes the 21st member State to join the Council of Europe.
Read more
A first for nature
SwitzerlandThe Bern Convention on the Conservation of European Wildlife and Natural Habitats is adopted, the first international treaty to protect wildlife and the natural environment.
Read more
Seventh Secretary General takes office
AustriaFranz Karasek becomes the seventh Secretary General of the Council of Europe.
Read more
Pompidou Group tackles drug policy
FranceThe Pompidou Group, European cooperation platform to combat drug abuse and illicit trafficking in drugs, becomes an enlarged partial agreement of the Council of Europe.
Read more
Solidarity founded
PolandThe Solidarity Movement is founded in the Gdańsk shipyard under the leadership of Lech Wałęsa.
Read more
Court's sixth President
NetherlandsGérard J. Wiarda becomes the sixth President of the European Human Rights Court.
Read more
Martial law in Poland
PolandGeneral Jaruzelski's authoritarian regime imposes martial law in the Polish People's Republic.
Read more
Controlling movements of firearms between countries
All CountriesConvention on Control of the Acquisition and Possession of Firearms by Individuals enters into force.
Read more
Towards a Europe without the death penalty
FranceOpening of Protocol 6 to the European Convention on Human Rights abolishing the Death Penalty
Read more
Eighth Secretary General takes office
SpainMarcelino Oreja Aguirre becomes the eighth Secretary General.
Read more
Towards a Europe without the death penalty
All CountriesProtocol No. 6 to the European Human Rights Convention makes abolition of the death penalty a precondition for Council of Europe membership.
Read more
Court's seventh President
NorwayRolv Ryssdal becomes the seventh President of the European Human Rights Court.
Read more
Privacy protected
All CountriesThe Council of Europe leads the way in protecting privacy when personal data is processed with what is still the only binding international convention with a potential worldwide remit.
Read more
Making sport safe
All CountriesEuropean Convention on Spectator Violence and Misbehaviour at Sports Events enters into force, bringing in stricter rules for sports grounds and crowd control.
Read more
Fewer experiments on animals
FranceOpening of the Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes
Read more
Quick response to disasters guaranteed
FranceThe EUR-OPA agreement starts work to predict, prevent and co-ordinate responses to major natural and technical disasters.
Read more
Democracy close to home
All CountriesThe European Charter of Local Self-Government enters into force to guarantee democracy at local level and give local authorities power of decision. It is one of the earliest legal texts to promote subsidiarity - taking decisions closest to citizens.
Read more
San Marino joins
San MarinoSan Marino becomes the 22nd member State to join the Council of Europe.
Read more
Torture watchdog created
All CountriesThe European Convention against Torture & Inhuman and Degrading Treatment enters into force allowing specialised teams to visit places of detention throughout Europe.
Read more
Finland joins
FinlandFinland becomes the 23rd member State to join the Council of Europe.
Read more
First woman Secretary General takes office
FranceCatherine Lalumière becomes the ninth Secretary General and the first woman to hold the post.
Read more
Gorbachev's message to Europe
Russian FederationMikhail Gorbachev outlines his idea of ‘a common European home’ in a speech to the Parliamentary Assembly.
Read more
Fall of the Berlin Wall
GermanyThe East German government announces that all citizens of the German Democratic Repuplic can visit West Germany and West Berlin.
Read more
Spreading values worldwide
PortugalThe Lisbon-based Centre for Global Interdependence and Solidarity - more commonly known as the "North-South Centre" - is launched to spread universal values of human rights, democracy and the rule of law beyond the European continent.
Read more
Dictatorship ends in Romania
RomaniaNicolae Ceaușescu and his wife are shot in Târgoviște at the end of the Romanian revolution.
Read more
Václav Havel becomes President
Czech RepublicVáclav Havel is elected President of Czechoslovakia after the Velvet Revolution brings an end to one-party rule in the country.
Read more
Action against sports cheats
All CountriesThe Anti-Doping Convention enters into force, allowing countries to work together to efficiently fight doping in sport.
Read more
Václav Havel calls for courage to look at the stars
FranceVáclav Havel, President of Czechoslovakia, 1989-1992 and first President of the Czech Republic, 1993-2003, addresses the Parliamentary Assembly, saying ‘without dreaming of a better Europe we shall never build a better Europe.’
Read more
Help for constitutional standard-setting
FranceThe Venice Commission, formally known as the European Commission for Democracy through Law, is established as a consultative body of legal experts to help countries build the values of the Council of Europe into their constitutions.
Read more
Russia signals end of Soviet Union
Russian FederationThe Russian Soviet Federative Socialist Republic declares its sovereignty as a state, beginning constitutional reform in Russia.
Read more
Hungary joins - first from central and eastern Europe
HungaryHungary becomes the 24th member State, the first to join from central and eastern Europe.
Read more
Criminals can't profit from crimes
FranceThe Convention on Laundering, Search, Seizure and Confiscation of the Proceeds from Crime is opened for signature. It boosts co-operation in investigations and tracking down, seizing and confiscating the proceeds of crime.
Read more
End of one-party rule in Albania
AlbaniaAlbania's Communist party allows political pluralism.
Read more
Break-up of Yugoslavia begins
SloveniaSlovenia leaves the Yugoslav Federation.
Read more
Poland joins
PolandPoland becomes the 25th member State to join the Council of Europe.
Read more
Bulgaria joins
BulgariaBulgaria becomes the 26th member State to join the Council of Europe.
Read more
European Audiovisual Observatory launches
FranceThe European Audiovisual Observatory is created to collect and distribute information about the various audiovisual markets in Europe and their financing.
Read more
Czechoslovakia ceases to exist
Czechoslovakia splits into the Czech Republic and Slovakia
Read more
Estonia joins
EstoniaEstonia becomes the 27th member State to join the Council of Europe.
Read more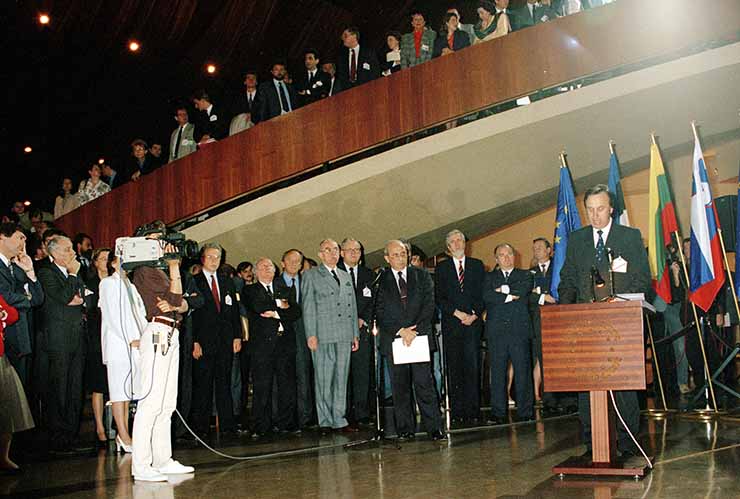
Lithuania joins
LithuaniaLithuania becomes the 28th member State to join the Council of Europe.
Read more
Slovenia joins
SloveniaSlovenia becomes the 29th member State of the Council of Europe.
Read more
Czech Republic joins
Czech RepublicThe Czech Republic becomes the 30th member State to join the Council of Europe.
Read more
Slovakia joins
Slovak RepublicSlovakia becomes the 31st member State to join the Council of Europe.
Read moreRomania joins
RomaniaRomania becomes the 32nd member State to join the Council of Europe.
Read more
Vienna Summit
AustriaFirst Summit of Heads of State and Government of the Council of Europe
Read more
Grassroots democracy given a voice
FranceThe Congress of Local and Regional Authorities of Europe is created to give a voice to towns, cities and regions.
Read more
Fighting racism and discrimination
FranceThe first meeting of the European Commission against Racism and Intolerance (ECRI). The Commission specialises in fighting all forms of racism, discrimination, xenophobia, anti-Semitism and intolerance.
Read more
Supporting film production
FranceThe European Convention on Cinematographic Co-Production enters into force and Eurimages is created to promote independent filmmaking with financial support for feature-length films, animation and documentaries.
Read moreTenth Secretary General takes office
SwedenDaniel Tarschys becomes the tenth Secretary General.
Read moreAndorra joins
AndorraAndorra becomes the 33rd member State to join the Council of Europe.
Read more
Latvia joins
LatviaLatvia becomes the 34th member State to join the Council of Europe.
Read more
New Human Rights building opened
FranceInauguration of the Human Rights Building, housing the European Human Rights Court, built by the British architect Lord Richard Rogers. Czech President Václav Havel attends.
Read more
Albania joins
AlbaniaAlbania becomes the 35th member State to join the Council of Europe.
Read more
Moldova joins
Republic of MoldovaMoldova becomes the 36th member State to join the Council of Europe.
Read more
Srebrenica massacre
Bosnia and HerzegovinaMore than 8,000 people are massacred in Srebrenica.
Read more
Ukraine joins
UkraineUkraine becomes the 37th member State to join the Council of Europe.
Read more
"The former Yugoslav Republic of Macedonia" joins
North Macedonia"The former Yugoslav Republic of Macedonia" becomes the 38th member State to join the Council of Europe.
Read more
USA becomes observer
United StatesThe United States of America is granted observer status.
Read more
Dayton Peace Accords
FranceThe Dayton Peace Accords end the Bosnian War.
Read more
Budapest Youth Centre opens
HungaryEuropean Youth Centre opens in Hungary.
Read more
Russia joins
Russian FederationThe Russian Federation becomes the 39th member State of the Council of Europe.
Read more
Croatia joins
CroatiaCroatia becomes 40th member State to join the Council of Europe.
Read more

Death Penalty ends
All CountriesThe very last execution takes place on the Council of Europe's territory. There have been none since.
Read more
Protecting rights in biomedicine
PortugalThe Oviedo Convention is opened for signature and becomes the only internationally binding treaty to protect human rights in the biomedical field. In 1998 it was amended to include a ban on human cloning.
Read more

National minorities protected against discrimination
All CountriesNational minorities get protection against discrimination with the entry into force of the Framework Convention for the Protection of National Minorities.
Read more
Regional or minority languages protected
All CountriesEurope's regional and minority languages get protection with the entry into force of the European Charter for Regional or Minority Languages.
Read more
Court's eighth President
GermanyRudolf Bernhardt becomes the eighth President of the European Human Rights Court.
Read more
Good Friday Agreement signed
United KingdomThe Good Friday Agreement is signed, bringing an end to 'The Troubles' in Northern Ireland
Read more
"The new Court"
FranceThe European Court of Human Rights' operation is streamlined with a protocol restructuring the control machinery. Since 1998 it has sat as a full-time court and individuals can apply to it directly.
Read more
Court's Ninth President
SwitzerlandLuzius Wildhaber becomes the ninth President of the European Human Rights Court.
Read more
Georgia joins
GeorgiaGeorgia becomes the 41st member State to join the Council of Europe.
Read more
Eleventh Secretary General takes office
AustriaWalter Schwimmer becomes the eleventh Secretary General.
Read more
First Human Rights Commissioner
SpainÁlvaro Gil-Robles becomes the first Human Rights Commissioner.
Read more
Children's rights in Court proceedings
All CountriesThe European Convention on the Exercise of Children’s Rights boosts children's protection, giving them the right to be heard and to express their views in court proceedings affecting them.
Read more
Discrimination banned
ItalyA general ban on discrimination comes in as a new protocol is opened for signature.
Read more
A tool for language learners
FranceEurope gets a common tool to measure language abilities thanks to the Common European Framework of Reference for Languages, launched at the beginning of the European Year of Languages.
Read more
Azerbaijan joins
AzerbaijanAzerbaijan becomes the 43rd member State to join the Council of Europe.
Read More
Armenia joins
ArmeniaArmenia becomes the 42nd member State to join the Council of Europe.
Read moreBosnia and Herzegovina joins
Bosnia and HerzegovinaBosnia and Herzegovina becomes the 44th member State to join the Council of Europe.
Read more
Death Penalty ban
LithuaniaThe Council of Europe adopts a protocol banning the death penalty in all circumstances
Read more
Corruption criminalised
All CountriesThe Criminal Law Convention on Corruption criminalises corrupt practices and improves international co-operation in their prosecution.
Read more
Serbia joins
SerbiaSerbia becomes the 45th member State to join the Council of Europe.
Read more
Protection against corruption
All CountriesThe Civil Law Convention on Corruption brings in international rules, asking governments to provide remedies in their domestic law for anyone who has suffered damage as a result of corruption.
Read more
Civil society representation boosted
FranceParticipatory status granted to international non-governmental organisations with the Council of Europe.
Read More
Georgia's Rose Revolution ends
GeorgiaPresident Eduard Shevardnadze resigns following the protests of the Rose Revolution, triggering presidential and parliamentary elections.
Read more
Combating cybercrime
All CountriesBudapest Convention - the first and only international legal treaty to tackle cybercrime - enters into force.
Read more
Twelfth Secretary General takes office
United KingdomTerry Davis becomes the 12th Secretary General.
Read more
Monaco joins
MonacoMonaco becomes the 46th member State to join the Council of Europe.
Read more
Ukraine's Orange Revolution ends
UkraineViktor Yushchenko is inaugurated as president of Ukraine.
Read more
Warsaw Summit
PolandThird Summit of 46 Heads of State and Government of the Council of Europe
Read more
Second Human Rights Commissioner
SwedenThomas Hammarberg becomes the second Human Rights Commissioner.
Read more
Serbia and Montenegro separate
SerbiaThe State Union of Serbia and Montenegro is dissolved.
Read more
Court's tenth President
FranceJean-Paul Costa becomes the 10th President of the European Human Rights Court.
Read more
EDQM building opens
FranceInauguration of the European Directorate for the Quality of Medicines and HealthCare (EDQM) building.
Read more
Montenegro joins
MontenegroMontenegro becomes the 47th member State to join the Council of Europe following its independence.
Read more
Stopping terrorists in their tracks
All Countries"A new convention bolsters the fight against terrorism with measures to criminalise public provocation, recruitment and training and to protect and compensate victims. "
Read more
Protecting children against sex abuse
SpainThe Lanzarote Convention is opened for signature, the first pan European treaty to make child sex abuse in the home, in the family or abroad a criminal offence.
Read more
Combating human trafficking
All CountriesA Convention to combat all forms of human trafficking comes into force, pledging assistance for victims, effective investigation and prosecution and stronger international co-operation.
Read more
The Agora building opens
FranceInauguration of the Agora, the Council of Europe's newest building.
Read more
Tackling terrorism financing
All CountriesThe Warsaw Convention, the first international treaty covering both prevention and control of money laundering and the financing of terrorism, comes into force.
Read more
Celebrating and promoting diversity
FranceThe Council of Europe launches its "White Paper on Intercultural Dialogue" as a blueprint for open and diverse societies based on human rights, democracy and the rule of law.
Read more
10,000 human rights judgments
FranceThe European Human Rights Court delivers its 10,000th judgment.
Read more
Thorbjørn Jagland is thirteenth Secretary General
NorwayThorbjørn Jagland takes office as the thirteenth Council of Europe Secretary General.
Read more
Efficient human rights protection
FranceProtocol No. 14 to the Convention enters into force, aiming to guarantee the long-term efficiency of the European Human Rights Court.
Read more
Making drugs safe
Russian FederationThe Medicrime Convention - aimed at stopping counterfeiting of medical products and similar crimes that threaten public health - is opened for signature.
Read more
Court's eleventh President
United KingdomSir Nicolas Bratza becomes 11th President of the European Human Rights Court.
Read more
Third Human Rights Commissioner
LatviaNils Muižnieks becomes the third Human Rights Commissioner.
Read more
Court's twelfth President
LuxembourgDean Spielmann becomes 12th President of the European Human Rights Court.
Read more
Secretary General re-elected
NorwayThorbjørn Jagland is re-elected, the first time a serving secretary general has secured a second term.
Read more
An end to violence against women
All CountriesThe Convention on preventing and combatting violence against women and domestic violence enters into force, with wide-ranging measures aiming to stop all forms of violence against women.
Read more
Stopping match-fixing
SwitzerlandThe Council of Europe launches a new Convention on the Manipulation of Sports Competitions to stop match-fixing and illegal sports betting operations.
Read more
Thirteenth Human Rights Court President
ItalyGuido Raimondi becomes thirteenth President of the European Human Rights Court.
Read more
A better approach to safety at sports events
FranceOpening of Convention on an Integrated Safety, Security and Service Approach at Football Matches and Other Sports Events
Read more
Saving Heritage from destruction
CyprusThe Convention on Offences relating to Cultural Property is opened for signature, the first international treaty to stop crimes that are destroying the world's cultural heritage such as theft, intentional damage, unlawful excavation and illegal sales.
Read more
Action against Organ Trafficking
All CountriesThe Convention against Trafficking in Human Organs enters into force.
Read more
Fourth Human Rights Commissioner - first woman
Bosnia and HerzegovinaDunja Mijatović becomes the Council of Europe's fourth Human Rights Commissioner - the first woman to hold the post.
Read more
Stronger links on human rights
FranceProtocol 16 to the European Human Rights Convention allows national courts to request advisory opinions on questions of principle.
Read more
Human Rights for an AI future
FranceCouncil of Europe sets up a taskforce to pinpoint the threats and opportunities of artificial intelligence for human rights.
Read more
Council of Europe reaches 70
All Countries70th Anniversary of the founding of the Council of Europe.
Read more














